Just hours before the end of the third month of the year, the Cuban government has again broken its word: in February the state company BioCubaFarma promised that "in March" it would deliver to the World Health Organization (WHO) the documentation necessary for the homologation of the anti-Covid vaccines produced on the island, but there has yet to be any official announcement about it.
The Cuban authorities have cited two dates since September to commence the approval process for these drugs, and Havana's new failure to follow through has delayed for more than half a year compliance with the protocol and the ensuing ability to use the injectable globally.
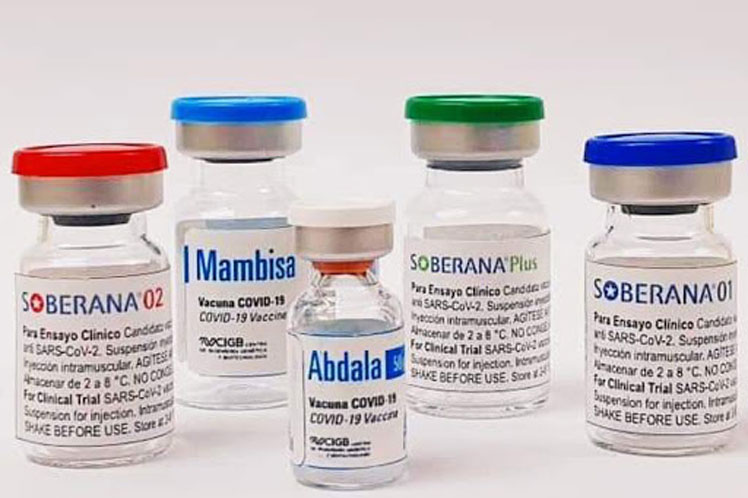
The president of BioCubaFarma, Eduardo Martínez, explained in mid-February that the documentation on the Abdala vaccine, one of the three developed by Cuba, and the first that was to pass the WHO's prequalification process, includes the delivery of information on clinical studies and other data related to production conditions.
At the end of January the authorities announced that they would deliver the documentation in the first half of February. In September 2021 they said the same thing. March was the third date they cited by which they would comply with the procedure.
According to Martínez, communications with representatives of international health agencies have been "continuous," and it is in Havana's best interest for Cuban vaccines to receive international approval, EFE said.
WHO approval would facilitate the international marketing of such vaccines, which could become a source of revenue for a country undergoing a serious economic crisis.
However, on March 31, the drug evaluation document that the WHO makes public stated that Abdala's Expression of Interest (EOI) procedure is still under review, while that for Sovereign 01, Sovereign 02 and Sovereign Plus are "still in need of information, strategies and the delivery schedule."
DIARIO DE CUBA combed social media and the official sites of the various state agencies involved in the procedure, including the Ministry of Public Health (MINSAP), and none refer to the postponement or impending completion of the required procedures.
Despite the fact that in 2020 the Cuban government announced that it would develop accessible and safe vaccines for the developing world, thus far only its political allies are using them: Venezuela, Nicaragua, Iran, Syria, Saint Vincent and the Grenadines, and Vietnam, although the government states that it has been negotiations with 15 other developing countries since 2021, and Mexico has announced that it will use them. A few weeks ago a contingent of officials and scientists in charge of creating the vaccines travelled to Italy and France.
In the first of these countries, Vicente Vérez, general director of the Finlay Institute of Vaccines confessed that the purpose of the visit was "to find a way for, at a certain time, our vaccines to be used in Italy, and the rest of Europe, on the one hand; and, in collaboration with Italy, reach children from Africa and other countries."
The real intention behind Havana's vaccine strategy seems to be to bypass all the protocols. This was evidenced when in late January the Government organized a virtual forum to promote what it termed a "life-saving package" for the Global South that included export of its life-saving vaccines through bilateral agreements with other States, the transfer of technology "where possible" for production in low-income countries, and the export of doctors for the distribution of these drugs in the countries concerned.
Using Progressive International (an organization comprised of leftist organizations and figures around the world, created by former Democratic presidential candidate Bernie Sanders and former Greek finance minister Yanis Varoufakis), as a sponsor, the government launched its proposal at a press conference with the authorities of its biopharmaceutical industry and the MINSAP.
The officials promised that, "despite the US embargo, Cuba has sufficient funding, including from the Central American Bank for Economic Integration, to produce the 200 million doses" of its vaccines.
They also offered "solidarity prices" for these injectables, the transfer of technology "where possible, for production in low-income countries," and the "expansion of medical brigades to build medical capacity and training" for the "export of the Cuban vaccination model" to countries that request it.
At the meeting, Olga Lidia Jacobo-Casanueva, director of the Center for State Control of Medicines and Medical Devices (CECMED), demanded: "Regulatory agencies must facilitate access and eliminate unnecessary regulatory barriers (...) expediting and enabling other countries to provide vaccination coverage like that achieved in Cuba."
This was reiterated by Ileana Morales Suárez, director of Scientific and Technological Innovation at the MINSAP: "The exclusionary approach imposed by the prevailing neoliberalism, on the production and acquisition of vaccines must cease," she said.
Gerardo Guillén, director of Biomedical Research at the Center for Genetic Engineering and Biotechnology (CIGB), said that to buy Cuban vaccines, the Ministries of Health of other countries can convey their interest in the drugs or do so via diplomatic channels.
"Today the productive capacity we have could be oriented at the whole world, because we can achieve extensive immunization coverage in Cuba. But for this we need agreements that allow us to finance it. If we could mobilize financial resources from other sources, this would be more beneficial for poor countries, because we could engage in triangular cooperation," he stressed.
With the pandemic petering out, health authorities around the world beginning to treat Covid-19 like a seasonal flu, and vaccination coverage reaching 58.3% of the planet's people, Havana seems to have missed its chance play a role as a savior of humanity.